Uncontrolled reactions cause the detonation of an atomic bomb. These chain reactions can be controlled or uncontrolled.Ĭontrolled reactions occur in a nuclear reactor to generate electrical energy. Slow neutrons are more likely to hit the nucleus of another fuel atom. However, fast neutrons can become slow neutrons when they collide with particles in a moderator. Neutrons aresuitable projectiles to hit the nucleus because they have no electrical charge, and the atomic nucleus does not reject. This atomic nucleus fission and, in turn, releases more fast neutrons giving the opportunity to repeat the process. What Is a Chain Reaction in Nuclear Fission?Ī chain reaction in nuclear fission is the repetitive process by which neutrons released in a first nuclear fission bump into other atoms and produce additional fissions. For this reason, the collision with a single neutron is enough to destabilize and break the entire structure. 
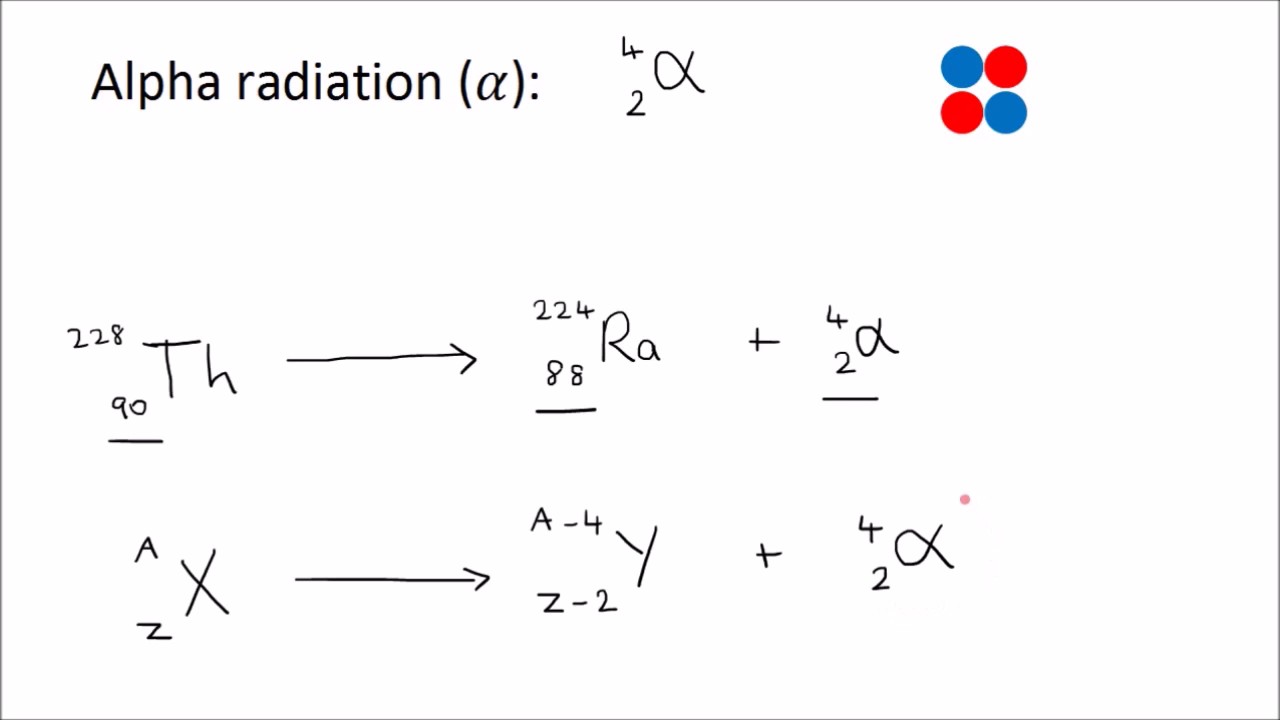
Having so many positively charged protons, the nucleus has a hard time maintaining the force bonds to hold them together. For example, the isotope of uranium-235 is more unstable than natural uranium. Isotopes are atoms of the same element but with different numbers of neutrons. Uranium and plutonium isotopes are ideal for this purpose because they arecumbersomey, with many positively charged protons in the nucleus. The material used as a nuclear fuel has a very unstable atomic structure. What Is the Chemical Element Used in a Nuclear Fission Reaction? Controlling the number of neutrons inside the reactor, they control its power. Nuclear reactors use free neutrons to undergo fission reactions. Nuclear fission can occur when a nucleus of a heavy atom captures a neutron or naturally without any interaction due to isotope instability. The energy resulting from a fission reaction is in the form of heat. “c” is a constant, the speed of light, which is 299,792,458 m / s 2. The missing mass is converted into energy according to Einstein's equation: In each fission process, there is a loss of the total mass: the sum of the masses of the fission products is less than the original mass of the atom. Nuclear Fission Energy and Einstein's Equation The reactions that take place in the Sun are of nuclear fusion. These fragments are fission products that, having changed their proton composition, are different chemical elements.įission should not be confused with nuclear fusion, which is a way of obtaining energy from the fusion of two light atoms. The purpose of a nuclear fission process is to alter this balance of forces, break this atomic force, and allow the nucleons to separate.Īfter the fission of the atomic nucleus, we obtain diverse fragments, two or three neutrons, and a significant quantity of energy emission. The nuclear force is a hundred times stronger than the electromagnetic force. The sub-particles of the atom are held together thanks to a nuclear force that holds them together. On the contrary, neutrons are neutral, meaning they have no charge. Protons have a positive charge therefore, they tend to repel each other. Depending on the element of the periodic table, the composition of these sub-particles varies. The nucleus of atoms comprises other smaller sub-particles: protons and neutrons. Through this process, we can obtain a large amount of energy. Some of them produce neutrons, called delayed neutrons, which contribute to the fission chain reaction.Nuclear fission is the physical- chemical reaction by which the nucleus of an atom is split into two smaller nuclei. 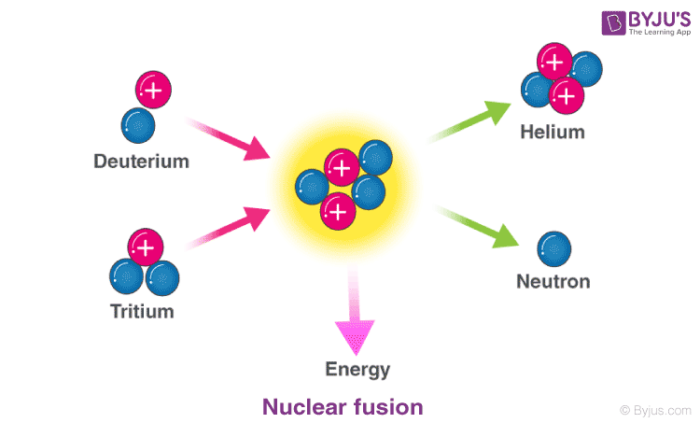
A critical chain reaction can be achieved at low concentrations of U-235 if the neutrons from fission are moderated to lower their speed, since the probability for fission with slow neutrons is greater.Ī fission chain reaction produces intermediate mass fragments which are highly radioactive and produce further energy by their radioactive decay. If the reaction will sustain itself, it is said to be "critical", and the mass of U-235 required to produced the critical condition is said to be a " critical mass". If an least one neutron from U-235 fission strikes another nucleus and causes it to fission, then the chain reaction will continue. If at least one neutron from each fission strikes another U-235 nucleus and initiates fission, then the chain reaction is sustained. Uranium-235 Fission Example Initiation of this processĮnergy From Uranium Fission Form of Energy ReleasedĮnergy of decay products of fission fragments Uranium-235 Chain Reaction Uranium-235 Fission Detailed example
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
